PVS: outcomes and pathophysiology
Overview of pulmonary vein stenosis
Pulmonary vein stenosis (PVS) is a rare and aggressive disease characterized by progressive lesions and results in secondary pulmonary hypertension and right heart failure. Infants and children with the severe progressive form of PVS have a 1 year survival of 50-60%. Despite advancements in surgical and interventional techniques, survival outcomes have plateaued over the last decade. There is an urgent need to increase our understanding around outcomes and risk factors associated with PVS to ensure advancement of clinical care for children with PVS.
PVS is typically broken down into 2 main clinical categories (Figure 1): Primary (or congenital) PVS or post repair PVS. Primary PVS typically encompasses children who develop PVS and have normally connected pulmonary veins and without prior surgery. Primary PVS can occur in the presence or absence of other congenital heart disease.
Post-repair PVS occurs in 10-15% of patients following repair of total anomalous pulmonary venous connections (TAPVC) and typically recurs within the first year after TAPVC repair (Karamlou T et al,. 2007). Risk factors for postoperative post-repair PVS include preoperative obstruction, mixed pulmonary venous connection, heterotaxy syndrome, and small pulmonary venous confluence (Jenkins K et al, 1993). In a large recent study in China, sutureless repairs for TAPVC decreased the risk of post-repair PVS in those with preoperative occlusion (Shi G, 2017).
Outcome studies for PVS have demonstrated similar survival curves over the past 2 decades. A collaborative PVS group involving the UK, Ireland and Sweden described clinical features, morphology and outcomes for their PVS cohort ( 2000-2007) , and they reported at 1 year survival of 55% and a 3 year survival of 49% (Seale AN, et al , 2009 ), with bilateral disease being a risk factor for poor survival. While the ability to generalize some of their data to contemporary cohorts is limited because the majority of patients (93%) did not undergo a sutureless repair (currently preferred surgical approach), a recent report from Boston Children's Hospital demonstrates a similar survival (Quinonez LG et al, 2015). Despite aggressive surgical, catheter based interventions and experimental therapies, this series reported a 1 year survival of 57% for primary PVS and survival at 1 year of 73% for post repair PVS. At 2 years, survival was 57% and 60%, respectively. Risk factors for decreased survival included younger age (<6months), lower weight (<3kg) and lower RVSP (<75% systemic).
PVS is typically broken down into 2 main clinical categories (Figure 1): Primary (or congenital) PVS or post repair PVS. Primary PVS typically encompasses children who develop PVS and have normally connected pulmonary veins and without prior surgery. Primary PVS can occur in the presence or absence of other congenital heart disease.
Post-repair PVS occurs in 10-15% of patients following repair of total anomalous pulmonary venous connections (TAPVC) and typically recurs within the first year after TAPVC repair (Karamlou T et al,. 2007). Risk factors for postoperative post-repair PVS include preoperative obstruction, mixed pulmonary venous connection, heterotaxy syndrome, and small pulmonary venous confluence (Jenkins K et al, 1993). In a large recent study in China, sutureless repairs for TAPVC decreased the risk of post-repair PVS in those with preoperative occlusion (Shi G, 2017).
Outcome studies for PVS have demonstrated similar survival curves over the past 2 decades. A collaborative PVS group involving the UK, Ireland and Sweden described clinical features, morphology and outcomes for their PVS cohort ( 2000-2007) , and they reported at 1 year survival of 55% and a 3 year survival of 49% (Seale AN, et al , 2009 ), with bilateral disease being a risk factor for poor survival. While the ability to generalize some of their data to contemporary cohorts is limited because the majority of patients (93%) did not undergo a sutureless repair (currently preferred surgical approach), a recent report from Boston Children's Hospital demonstrates a similar survival (Quinonez LG et al, 2015). Despite aggressive surgical, catheter based interventions and experimental therapies, this series reported a 1 year survival of 57% for primary PVS and survival at 1 year of 73% for post repair PVS. At 2 years, survival was 57% and 60%, respectively. Risk factors for decreased survival included younger age (<6months), lower weight (<3kg) and lower RVSP (<75% systemic).
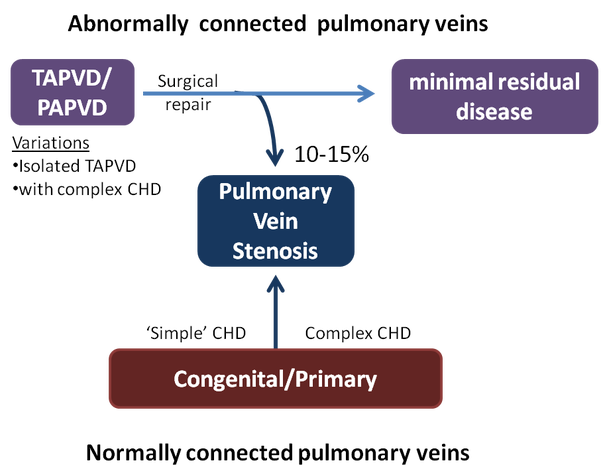
Figure 1. Clinical categories of PVS Abnormally connected pulmonary veins can occur in isolation or in the presence of complex CHD. After repair, 10-15% of patients will develop progressive pulmonary vein stenosis. Patients with normally connected pulmonary veins (who have had no surgery on their pulmonary veins) can also develop pulmonary vein stenosis- this can occur in the setting of associated simple congenital heart lesions (e.g septal defects) or more complex congenital heart lesions- such as hypoplastic left heart syndrome. TAPVD, total anomalous pulmonary venous drainage; PAPVD, partial anomalous pulmonary venous drainage
PVS that occurs in the very premature is being increasingly recognized as an important subgroup of patients. Initial reports highlighted the high number of children with PVS were born premature. A recent multicenter retrospective study identified risk factors for PVS in ex-premature infants (Mahgoub, L et al, 2017). This cohort included children with a median gestational age of 28 weeks (22-36) who required a median of 5 echocardiograms prior to a diagnosis of PVS at the median age of 6.5 months. In this ex-premature cohort, 74% had a diagnosis of bronchopulmonary dysplasia and 67% had a diagnosis of pulmonary hypertension. Survival was 77% at 1 year and 55% at 2, 5 and 10 years. Risk factors echo those from previous cohort studies greater than 3 pulmonary veins involved, bilateral PVS and earlier age at diagnosis were associated with decreased survival.
PVS: etiology and disease mechanisms
The etiology or inciting trigger for PVS remains elusive. PVS is frequently associated with other cardiac defects and has a heterogenous presentation. Congenital PVS is rare (~0.5%) and only one genetic loci (with no candidate gene identified) has been associated with congenital PVS and abnormal lymphatic development ( VandeLaar I, et al, 2009). However, no other genetic associations have been identified for PVS , and likely points to a multifactorial disease process. Retrospective studies have found associations with prematurity and the presence of necrotizing enterocolitis (Drossner DM et al, 2008; Heching HJ et al, 2014), however these association are correlative and not causative. Associations between pulmonary veins and external compression from other thoracic structures have also been proposed to increase the risk of stenosis ( Kotani Y et al, 2015).
Histological analysis of pulmonary veins from children with pulmonary vein stenosis demonstrates luminal occlusion with neointimal lesions. These lesions may be focal, diffuse or in advanced disease- the affected pulmonary vein may be hypoplastic or atretic (Figure 1a-c). Neointimal lesions are characterized by myofibroblast-like cells with extracellular deposition ( Sadr IM et al 2000, Kato H et al, 2014). Immunohistochemistry of endstage lesions suggest that some tyrosine kinase receptors are activated, but the significance of this remains unknown (Riedlinger WF et al 2006). In addition, a surgical porcine model of PVS demonstrates similar myofibroblast and extracellular matrix accumulation and is associated with elevated TGF-β signaling and activation of Angiotensin II pathways ( Kato H et al 2014; Zhu J et al, 2015). Despite potentially different etiological events within different categories of PVS, common final signaling leads to myofibroblast deposition. This suggests that therapies targeting myofibroblast deposition might have a beneficial effect across different PVS subtypes. Research is being done to more fully understand neointimal formation in pulmonary vein stenosis.
Histological analysis of pulmonary veins from children with pulmonary vein stenosis demonstrates luminal occlusion with neointimal lesions. These lesions may be focal, diffuse or in advanced disease- the affected pulmonary vein may be hypoplastic or atretic (Figure 1a-c). Neointimal lesions are characterized by myofibroblast-like cells with extracellular deposition ( Sadr IM et al 2000, Kato H et al, 2014). Immunohistochemistry of endstage lesions suggest that some tyrosine kinase receptors are activated, but the significance of this remains unknown (Riedlinger WF et al 2006). In addition, a surgical porcine model of PVS demonstrates similar myofibroblast and extracellular matrix accumulation and is associated with elevated TGF-β signaling and activation of Angiotensin II pathways ( Kato H et al 2014; Zhu J et al, 2015). Despite potentially different etiological events within different categories of PVS, common final signaling leads to myofibroblast deposition. This suggests that therapies targeting myofibroblast deposition might have a beneficial effect across different PVS subtypes. Research is being done to more fully understand neointimal formation in pulmonary vein stenosis.
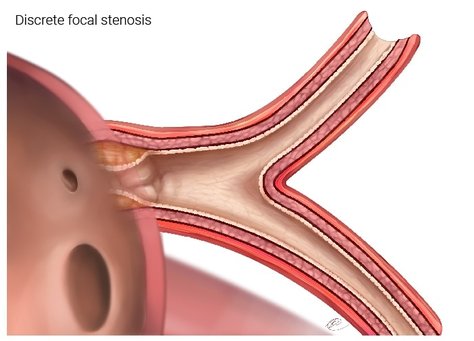
Figure 2a. This shows a pulmonary vein that has a discrete focal stenosis right at the junction of the pulmonary vein and the left atrium. The rest of the vein can have minimal or no disease. This type of disease can be addressed by surgical or catheter based interventions.
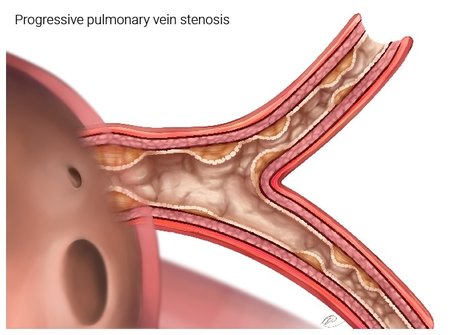
Figure 2b. This depicts a pulmonary vein that has more extensive disease. The areas of stenosis have extended up into the veins that are in the lung tissue (parenchyma), in addition to being close to the left atrium. This type of disease will be harder to treat and if spreads to more than one vein, can result affect lung function.
Surgery can only address the disease that is close to the left atrium. Blood can still flow in this vein, but the flow will be reduced.
Surgery can only address the disease that is close to the left atrium. Blood can still flow in this vein, but the flow will be reduced.
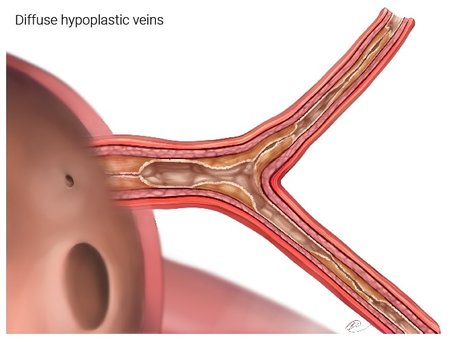
Figure 2c. This depicts a pulmonary vein that is severely affected by disease. Blood has very hard time draining back into the left atrium and it is hard for the right heart to pump blood into this part of the lung.
Myofibroblasts: potential sources of cells
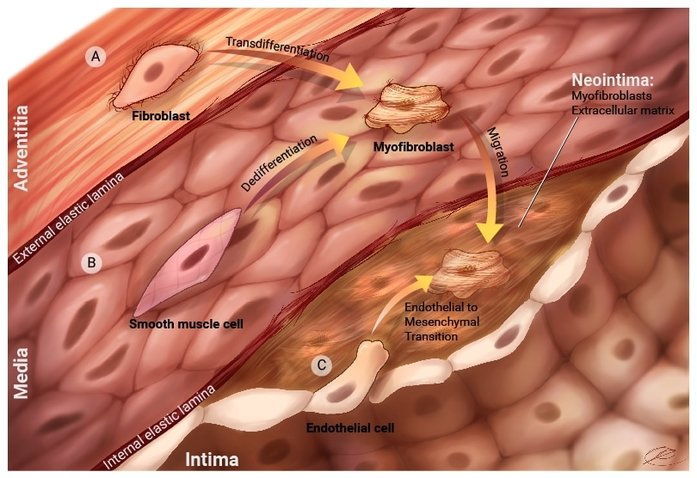
Myofibroblasts are the central cell type seen in PVS lesions. These cells take on secretory functions, migratory functions and have contractile phenotype. There are many potential sources of myofibroblast cells within vascular pathology. Most commonly, myofibroblasts can come from adventitial fibroblasts that proliferate and differentiate into myofibroblasts. These can then migrate to different layers within the vessel wall. Smooth muscle cells or pericytes can also differentiate into myofibroblast-like cells due to local signaling cues. Endothelial cells can differentiate into myofibroblast cells via a process termed endothelial-to-mesenchymal transition (EndMT). EndMT causes cells loss their cell-cell contacts, loss endothelial specific cell markers and gain fibroblast-like markers. Lastly, circulating progenitors may contribute to myofibroblast deposition (Figure 2). Development of therapies targeting any of these potential sources of myofibroblasts may help to slow the progression of disease and improve outcomes for children with PVS.
FIgure 3. Potential sources of cells are a) fibroblasts from the adventitia. These cells can proliferate in response to different signals and they can turn into myofibroblasts that are capable of migration into the neointima. b) Smooth muscle cells can be stimulated by different signaling molecules to turn into myofibroblasts that can migrate into the neointima. c) Endothelial cells can also undergo a specialized process called Endothelial-to-mesenchymal transition. Endothelial cells will loss specific cell markers and take on myofibroblast properties. These cells can then move into the neointima. There are potential other sources of cells , such as circulating progenitors or proliferating pericytes. Research is being done to more fully understand how the neointima forms in pulmonary vein stenosis.